 
The rarity, artistry, and tales associated with natural diamonds are what enhance their brightness. The only substance that can scratch a diamond is another diamond. Natural diamonds are incredibly durable and have a Mohs hardness rating of 10, making them resistant to scratching and wear. These factors are internationally recognised standards used to assess and grade diamonds. The value and quality of a natural diamond are determined by the 4Cs: Carat weight, Color, Clarity, and Cut. These imperfections can range from tiny crystals to fractures, and their unique patterns can be used to identify individual diamonds. These are the birthmarks that nature leaves within the gemstone. For instance, chemical elements such as nitrogen, sulphur, and boron can colour diamonds in shades of yellow, green and blue.Īlmost all natural diamonds contain tiny imperfections or inclusions. 
These colours are created when trace elements interact with the carbon atoms during the diamond's creation. While diamonds are often associated with the traditional white colour, they come in a wide array of natural hues - but only 1 in 10,000 diamonds is a fancy colour. Thus, wearing a diamond is nothing short of wearing a piece of ancient history. Most natural diamonds are estimated to be billions of years old, dating back to the formation of the Earth's crust. Each region produces diamonds with distinct characteristics, contributing to their unique beauty and desirability. Major diamond-producing countries include Russia, Botswana, Canada, DR Congo, and South Africa. 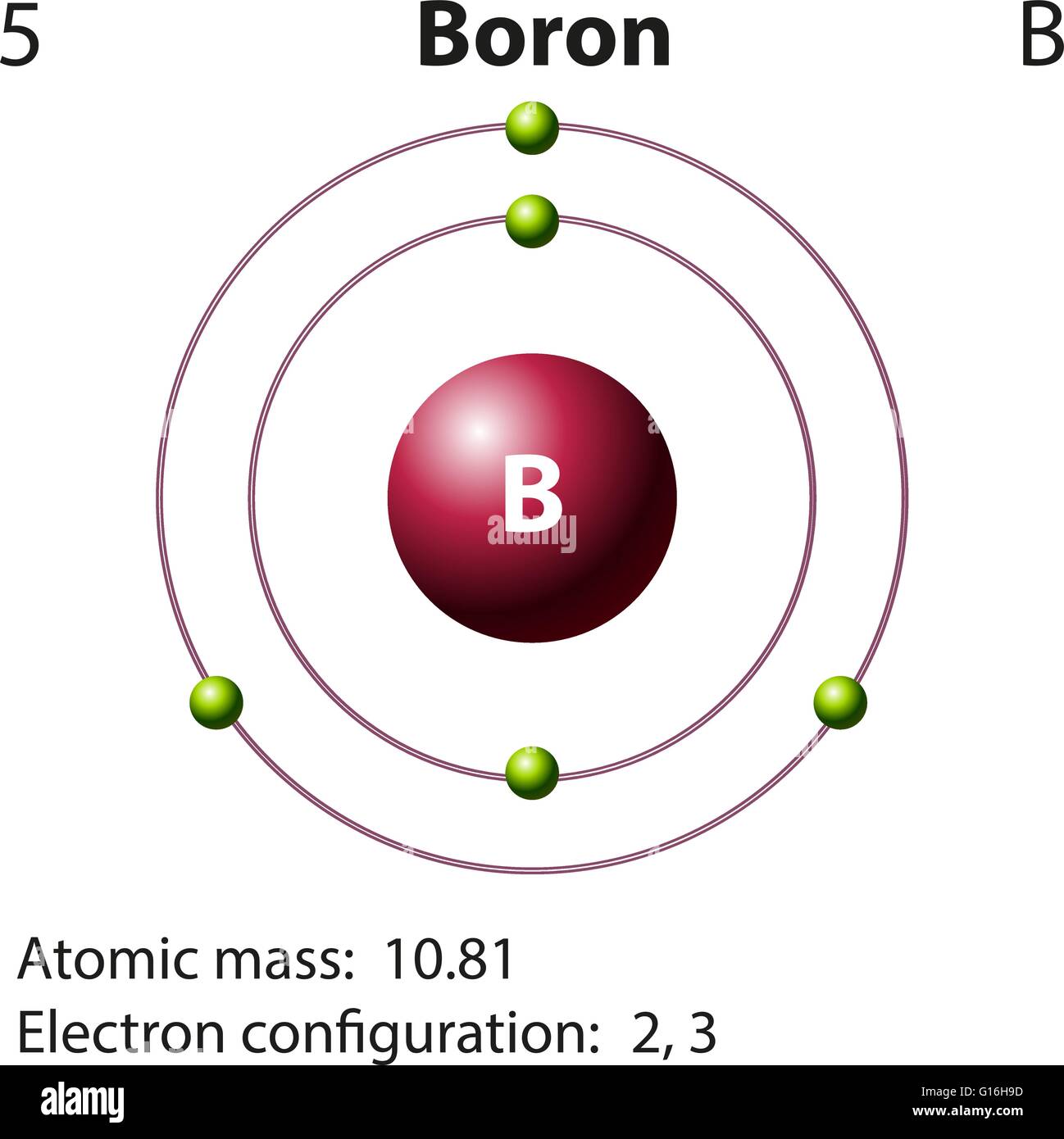
Natural diamonds are sourced from numerous geological locations around the world. This rarity adds to their value and allure. Only about one in a million diamonds mined worldwide are of gemstone quality. Although only traces are required, it is an essential plant nutrient.Despite their popularity, natural diamonds are rare. Boron-containing organic antibiotics are known. Borates have low toxicity in mammals (similar to table salt) but are more toxic to arthropods and are occasionally used as insecticides. Consensus on it as essential for mammalian life is lacking. The intersection of boron with biology is very small. Natural boron is composed of two stable isotopes, one of which ( boron-10) has a number of uses as a neutron-capturing agent. A few boron-containing organic pharmaceuticals are used or are in study. A small amount is used as a dopant in semiconductors, and reagent intermediates in the synthesis of organic fine chemicals. As sodium perborate, it is used as a bleach. Borosilicate glass is desired for its greater strength and thermal shock resistance than ordinary soda lime glass. The next leading use is in polymers and ceramics in high-strength, lightweight structural and heat-resistant materials. About half of all production consumed globally is an additive in fiberglass for insulation and structural materials. The primary use of the element itself is as boron filaments with applications similar to carbon fibers in some high-strength materials.īoron is primarily used in chemical compounds. Several allotropes exist: amorphous boron is a brown powder crystalline boron is silvery to black, extremely hard (about 9.5 on the Mohs scale), and a poor electrical conductor at room temperature. Industrially, the very pure element is produced with difficulty because of contamination by carbon or other elements that resist removal. The largest known deposits are in Turkey, the largest producer of boron minerals.Įlemental boron is a metalloid that is found in small amounts in meteoroids but chemically uncombined boron is not otherwise found naturally on Earth. These are mined industrially as evaporites, such as borax and kernite. It is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals. It constitutes about 0.001 percent by weight of Earth's crust. Boron is synthesized entirely by cosmic ray spallation and supernovae and not by stellar nucleosynthesis, so it is a low-abundance element in the Solar System and in the Earth's crust. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
